The 2026 CMS fee schedule digital health changes amount to a structural reset, not a rate adjustment. The CY 2026 Physician Fee Schedule (CMS-1832-F), effective January 1, 2026, rewrote the economic conditions for an entire category of remote monitoring and digital therapeutic programs.
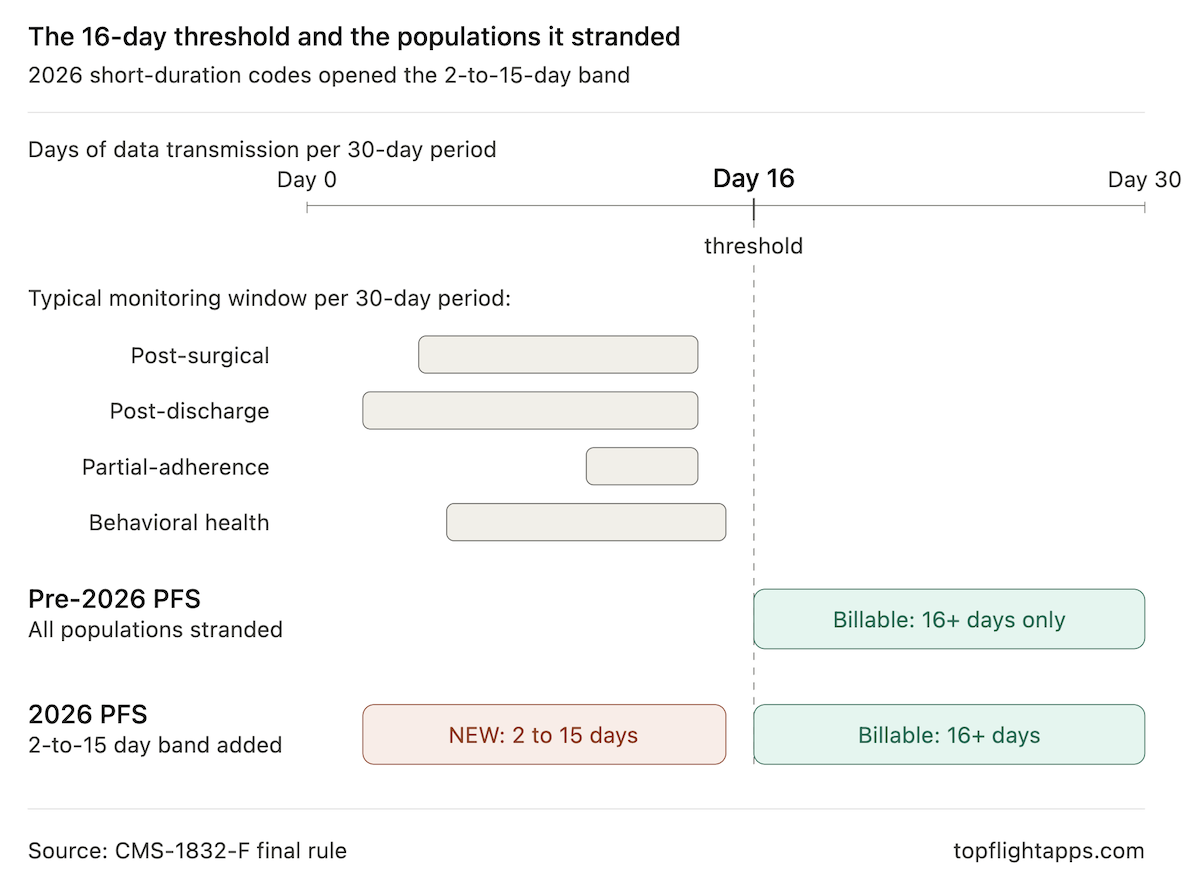
The 16-day-per-month remote physiologic monitoring (RPM) data transmission requirement made sense for chronic disease management, where a patient is monitored indefinitely. It was the wrong rule for most of the genuinely valuable monitoring scenarios:
- a patient recovering from a knee replacement who needs 10 days of post-surgical oversight
- a behavioral health patient engaging with a digital therapeutic for three weeks
- a post-discharge CHF patient in the critical fortnight after hospitalization
These patients had real clinical need. Their care teams were doing real clinical work. They simply didn’t hit the threshold, which meant no reimbursement, which meant no business model.
The new framework changes that. New short-duration codes bill for 2 to 15 days of data transmission. New 10-minute management codes bill for clinical review time that doesn’t reach the 20-minute threshold. DMHT reimbursement now covers ADHD digital therapeutics, with CMS signaling further expansion.
And buried in the rulemaking record is something more significant than any single code change: CMS asked stakeholders how it should value physician work when software drives clinical decisions, and indicated those questions will return in future rulemaking. Software-generated clinical data is becoming a billable input. The teams building for that reality now will be 12 to 18 months ahead of those who aren’t.
This piece translates the 2026 changes into product and architecture decisions. Not billing guidance for practice managers, but a builder’s briefing on what changed, which patient populations it unlocks, what your app needs to generate and log, and what the FDA/CMS coupling means for product decisions you’re making today.
What do builders need to know about the 2026 CMS fee schedule?
The CY 2026 Medicare Physician Fee Schedule (CMS-1832-F) broke the 16-day RPM data transmission floor with new short-duration codes (CPT 99445, 99470, 98984, 98985, 98986, 98979), expanded DMHT reimbursement to ADHD digital therapeutics, and added APCM behavioral health add-on codes (G0568, G0569, G0570). The structural shift for product teams is that CMS now treats software-generated clinical data as a verifiable billing input, which makes audit-trail architecture, FDA clearance status, and individualized documentation prerequisites for revenue, not optional polish.
Key Takeaways
- Episode-based monitoring is now reimbursable. The new 2 to 15 day device supply codes and 10 to 19 minute management codes make post-surgical, post-discharge, and behavioral health episodes billable for the first time, opening four patient populations that were previously economically unviable.
- Software is now a billable clinical input, not just a log. CMS’s framing under the 2026 codes treats platform-generated data as evidence of a clinical service. Tamper-evident logging, individualized documentation, and FDA-cleared device status are billing prerequisites, enforced by audit.
- FDA classification decisions today determine CPT eligibility tomorrow. The gap between FDA codification and CMS reimbursement has compressed to roughly 16 months. Picking a 510(k) classification is also picking a future revenue model, with sleep-disturbance, GI, and fibromyalgia digital therapeutics next in line.
Table of Contents
- What Actually Changed: The 2026 Codes in Plain Language
- Patient Populations Unlocked
- The SaMD Verification Concept: Why This Is More Than a Code Change
- What Your App Needs to Generate: The Technical Billing Requirements
- The FDA/CMS Coupling: Why SaMD Classification Decisions Made Today Have Billing Consequences in 18 Months
- The Business Model Math
- Builder Action List: Five Product Decisions the 2026 Rule Should Trigger
- Why Choose Topflight for Reimbursement-Enabled Digital Health Development
What Actually Changed: The 2026 Codes in Plain Language
The 2026 Physician Fee Schedule made three structural changes that matter for builders. It broke the 16-day RPM/RTM data transmission floor with a new family of 2 to 15 day device supply codes and 10 to 19 minute management codes. It expanded DMHT reimbursement to ADHD digital therapeutics.
And it introduced Advanced Primary Care Management (APCM) behavioral health add-on codes that let care management platforms layer behavioral health integration (BHI) and Collaborative Care Model (CoCM) services on top of an existing APCM base code.
Here is what changed at the code level.
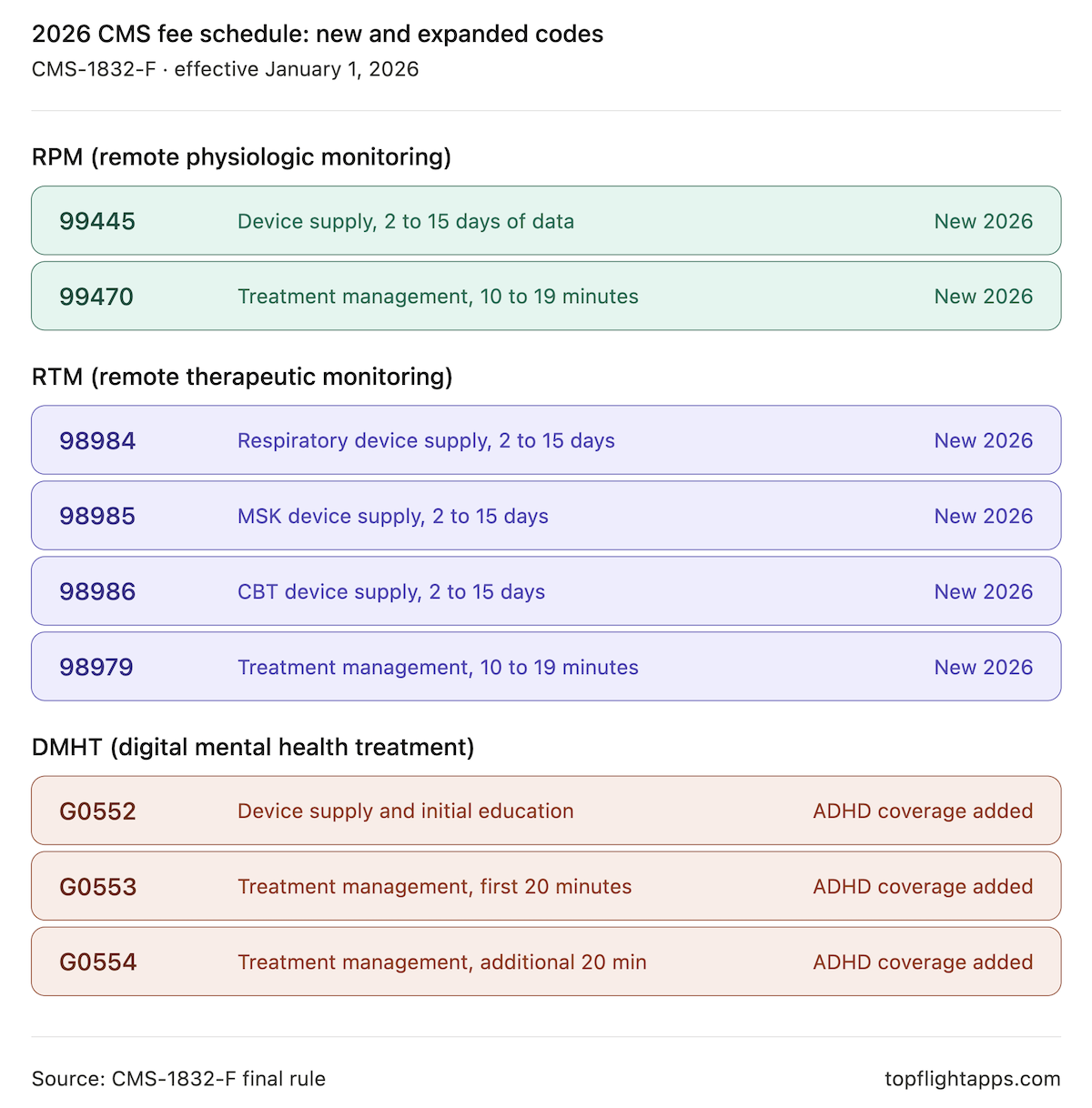
RPM, New and Revised Codes
| Code | What It Covers | Key Change |
| 99445 (new) | RPM device supply, 2 to 15 days of data in a 30-day period | New code; valued same as 99454 and mutually exclusive with it |
| 99454 (revised) | RPM device supply, 16 to 30 days of data | Descriptor updated; rate unchanged |
| 99470 (new) | RPM treatment management, 10 to 19 minutes per calendar month | New code at 0.31 work RVUs, half of 99457; mutually exclusive with 99457 |
| 99457 (unchanged) | RPM treatment management, 20+ minutes per month | Meets the interactive communication requirement (at least one live interaction) |
| 99458 (unchanged) | RPM additional 20-minute management increments | Unchanged |
The new 2026 RTM CPT codes mirror RPM in structure. The final rule added 2 to 15 day device supply codes for three monitoring domains (respiratory, musculoskeletal, cognitive behavioral therapy) and a parallel 10-minute management code:
| Code | What It Covers | Key Change |
| 98984 (new) | RTM respiratory device supply, 2 to 15 days | New code; mirrors 98976 |
| 98985 (new) | RTM MSK device supply, 2 to 15 days | New code; mirrors 98977 |
| 98986 (new) | RTM CBT device supply, 2 to 15 days | New code; mirrors 98978 |
| 98979 (new) | RTM treatment management, 10 to 19 minutes per month | New code at 0.31 work RVUs, parallel to 99470 |
| 98977 to 98981 (unchanged) | RTM 16+ day device supply and 20+ minute management | Unchanged |
DMHT, 2026 Expansion
The DMHT framework for 2026 digital therapeutics, originally established in CY 2025 for FDA-cleared computerized behavioral therapy devices under 21 CFR 882.5801, now extends to FDA-cleared digital therapy devices for ADHD under 21 CFR 882.5803:
| Code | What It Covers | Key Change |
| G0552 | DMHT device supply and initial patient education | Now covers FDA-cleared ADHD devices; contractor pricing remains |
| G0553 | DMHT treatment management, first 20 minutes per month | ADHD device coverage added |
| G0554 | DMHT treatment management, additional 20-minute increments | ADHD device coverage added |
APCM Behavioral Health Add-Ons
G0568 (initial CoCM), G0569 (subsequent CoCM), and G0570 (general BHI) are billable only when an APCM base code (G0556, G0557, or G0558) is reported for the same patient in the same month. They create a billing pathway for integrating behavioral health into APCM programs at primary care practices and within FQHC rural health clinic billing workflows, supporting patients with co-occurring chronic and behavioral conditions.
What CMS Declined but Signaled
The agency did not extend DMHT to GI conditions or fibromyalgia in 2026, though both are under consideration for future rulemaking. More significantly, the CMS SaaS AI RFI asked how CMS should value physician work when software drives clinical decisions, and how software costs should enter the practice expense methodology. In the PFS final rule 2026, CMS said it would address those questions in future rulemaking.
The next one to two cycles will work through how AI-enabled clinical tools are reimbursed. These signals fit within the broader DHHS digitization initiative pushing AI in remote patient monitoring and other clinical software toward reimbursement-grade infrastructure.
Patient Populations Unlocked
The 16-day rule didn’t just constrain billing. It constrained which patients got remote monitoring at all. Programs were built around the patients who would generate enough data to be reimbursable, which biased the entire category toward chronic disease management with indefinite monitoring horizons.
What short duration RPM billing 2026 unlocks is a different set of patient populations and a different unit of clinical work: the episode.
Post-Surgical Recovery
A patient discharged after total knee arthroplasty needs vital signs, pain scores, and mobility data for the first one to two weeks at home. Same-day discharge orthopedic protocols are now standard, and the critical monitoring window typically runs 4 to 14 days.
Under the old code structure, none of this clinical work was reimbursable because it ended before day 16. Under the new structure, a 10-day post-op program bills cleanly, with management time captured at the 10-minute increment when a clinician reviews readings and checks in once or twice during the episode.
Post-Discharge Transition Care
The 14 days after a CHF, COPD, or pneumonia hospitalization are when readmission risk is highest and clinical attention has the most leverage. Programs designed around this window now have a billing pathway that matches the clinical reality, instead of being forced into a 30-day chronic-care framing that doesn’t fit the episode.
Behavioral Health Engagement
Most digital therapeutics are designed for finite engagement arcs:
- a six to eight week CBT program for insomnia
- a multi-week ADHD intervention
- a structured course of behavioral activation for depression
The new RTM device codes (98984, 98985, 98986) and the 10-minute management code (98979) accommodate these episodes natively. Combined with the DMHT framework’s expansion to ADHD devices, behavioral health builders, including those working through a companion app pharma partnership, now have two separate reimbursement pathways depending on whether the device qualifies as an FDA-cleared digital therapeutic or as remote therapeutic monitoring.
Partial-Adherence Chronic Disease
The most underestimated population. A diabetes or hypertension patient who consistently generates 10 to 14 days of readings per month was previously unreimbursable, which meant clinically real work went uncompensated and many chronic disease management app programs simply dropped non-adherent patients. Now that month is billable. The patient stays in the program, the data keeps flowing, and the care relationship continues instead of breaking.
Episode-based monitoring is finally economically viable. That changes which products get built.
The SaMD Verification Concept: Why This Is More Than a Code Change
Read the new codes carefully and a pattern emerges that the rule text never states directly. SaMD reimbursement CMS 2026 isn’t billing a clinician for time spent looking at a passive log of patient data. It is paying for clinical work that the software made possible by generating, validating, and contextualizing that data in the first place. The software is no longer documentation. It is a verified clinical input.
This is the conceptual shift builders need to internalize, because three consequences flow from it that shape product architecture.
Every Billed Claim Becomes an Audit Surface
When a practice bills 99445 for 12 days of data transmission, the question an auditor can ask is no longer “did the doctor look at the chart?” It is “can the platform prove that 12 distinct days of valid physiologic data were transmitted from a patient-attributed device, and that the data wasn’t manually entered or duplicated?” The answer has to be yes, and it has to be yes from the platform’s logs, not from the clinician’s notes. The minimum evidentiary stack:
- Tamper-evident timestamps for every reading
- Device attestation that confirms the source hardware
- Deduplication logic that prevents inflated day counts
- Per-day data validation that rejects partial or implausible readings
These are no longer nice-to-haves. Tamper-evident logging is the evidentiary basis for the claim.
Documentation Becomes Individualized
The 10-minute management codes (99470, 98979) require at least one live interactive communication per calendar month, and the platform has to capture what that communication was, when it happened, and what was discussed. CMS clarified in the final rule what now counts toward interactive communication time, provided the interactions meet CPT specifications:
- Real-time audio or video calls
- Secure messaging
- Asynchronous chat
- Bi-directional automated messaging
- AI-mediated prompts and responses
That expansion is meaningful, but it cuts both ways. Each interaction has to be individually logged with content, timestamp, and patient context. Generic “patient was contacted” entries don’t survive an audit. Specific, retrievable, per-patient communication records do.
FDA Clearance Is a Billing Prerequisite, Not a Marketing Point
This is most explicit in DMHT, where G0552, G0553, and G0554 require an FDA-cleared device. It is also implicit in the RPM device codes, which require automatic data transmission from a medical device that meets FDA’s definition of one. The line for when an RPM app becomes a medical device is the same line for when its data becomes billable. The classification pathway you choose for your product, and the regulatory timeline you pick, now directly determines which CPT codes your platform can support. A digital therapeutic that hasn’t completed 510(k) clearance under 21 CFR 882.5801 or 882.5803 cannot bill DMHT, regardless of how clinically effective it is.
The teams treating regulatory pathway as an architectural decision instead of a paperwork milestone, whether in digital therapeutics, SaMD platforms, or medical device companion app development, are the ones building reimbursable products.
What Your App Needs to Generate: The Technical Billing Requirements
Audit trail billing turns the SaMD verification concept into concrete data model requirements once you map them per code family. For teams revisiting their remote patient monitoring app development data model, here is the minimum each code requires your platform to produce and store, in a form that survives audit and supports clean claim submission.
RPM Device Data (99445, 99454)
For CPT 99445 RPM short duration billing, the platform must transmit data from an FDA-defined medical device for at least 2 distinct days within the 30-day period, and not more than 15. For 99454, the floor is 16 days. Each platform record needs:
- Patient ID, device serial number, and device-attestation token
- Per-reading timestamp (UTC, ISO 8601) and source flag distinguishing automatic from manual
- A “valid day” flag derived from at least one automatic, in-range reading per calendar day
- A monthly summary of data transmission days that automatically recommends 99445 versus 99454
RPM Management Time (99470, 99457, 99458)
Management codes are time-and-interaction codes. Per patient per calendar month:
- A running total of clinical staff time, attributable by user ID, with start and stop timestamps for each activity
- A log of at least one live interactive communication, including channel (call, video, secure message, async chat), duration, and a content summary
- Automatic flags when the cumulative total crosses the 10-minute and 20-minute thresholds, with a rule that prevents 99470 and 99457 from being recommended in the same month
RTM Data and Management (98984, 98985, 98986, 98979, 98980, 98981)
RTM mirrors RPM in structure but accepts patient-reported data alongside device-captured data. The platform needs the same day-count and time-tracking infrastructure, with two additions:
- A field identifying the monitoring domain (respiratory, MSK, or CBT) for the correct device code (98984, 98985, or 98986)
- Validation that distinguishes patient self-report from device transmission, since RTM permits self-report but the audit trail must show the source
DMHT (G0552, G0553, G0554)
DMHT carries an additional regulatory layer. The platform must record the FDA clearance number of the prescribed device, the date of patient education and device assignment (G0552), and per-month management time logs (G0553, G0554). Without the cleared-device identifier captured at the claim level, the codes are not billable. This is where EHR in medical billing integration matters: the device identifier needs to flow into the claim, not just live in the platform’s logs.
The FDA/CMS Coupling: Why SaMD Classification Decisions Made Today Have Billing Consequences in 18 Months
Once you accept that FDA clearance gates billing eligibility, the strategic implication is that today’s FDA classification decisions are tomorrow’s CMS reimbursement categories. The pattern is observable.
The first DMHT classification, 21 CFR 882.5801 for computerized behavioral therapy devices, was codified by FDA in December 2017. CMS finalized reimbursement for devices under that classification in the CY 2025 PFS, effective January 2025. The second classification, 21 CFR 882.5803 for ADHD digital therapy devices, was codified in the Federal Register in September 2024. CMS extended DMHT reimbursement to it in the CY 2026 final rule, effective January 2026. Roughly 16 months between codification and reimbursement.
The gap is compressing, and the next set of categories is already visible:
- 21 CFR 882.5705, digital therapy device to reduce sleep disturbance for psychiatric conditions. Established. No CMS reimbursement yet.
- GI digital therapeutics (codified under 21 CFR 876.5960). CMS declined inclusion in 2026 but said the door is open.
- Fibromyalgia digital therapeutics. Same status as GI: declined now, candidate for future rulemaking.
- AI-enabled clinical tools broadly. CMS deferred its SaaS and AI questions to future rulemaking, which is the strongest signal yet that an entirely new payment methodology is being worked out.
For builders, the takeaway is that SaMD classification billing consequences play out over 12 to 24 months: regulatory pathway and architectural decisions you make now determine which CPT codes your platform can access. Picking a 510(k) classification is also picking a future revenue model. (Is My Health AI a Medical Device? covers the SaMD classification framework in depth.)
The Business Model Math
The 2026 framework changes per-patient revenue arithmetic. Three illustrative scenarios at 2026 national non-facility averages, with the standard caveat that locality adjustments and your MAC apply.
The non-QP conversion factor 2026 moved from $32.35 to $33.40, a 3.26% increase that includes the temporary 2.5% statutory bump from the One Big Beautiful Bill Act, partially offset by a -2.5% efficiency adjustment that exempts time-based codes and new 2026 codes. The new short-duration RPM and RTM codes were valued using OPPS geometric mean cost, with RTM remaining on the New Technology List through 2030. That methodology change is the reason 2026 rates moved meaningfully higher than 2025.
Post-Surgical 10-Day Monitoring Episode
- 2025: zero reimbursement-eligible data, since 10 days falls below the 16-day threshold.
- 2026: CPT 99445 (~$52) plus CPT 99470 (~$26) = approximately $78 per patient per month.
A program enrolling 200 post-surgical patients per month moves from $0 to roughly $15,600 in monthly RPM revenue, or about $187,200 annualized.
Chronic Disease Program With 30% Partial-Adherence Patients
A 200-patient program where 60 patients transmit 10 to 14 days per month previously generated no device supply revenue in those months. CPT 99445 now captures them at the same rate as 99454. At ~$52 per patient, that is roughly $3,120 in recovered monthly revenue, about $37,440 annualized, with no clinical workflow change.
Maximally Stacked Patient
A patient with two or more chronic conditions and a co-occurring behavioral health diagnosis can support concurrent RPM, Chronic Care Management, and digital mental health treatment reimbursement 2026 billing, provided clinical time is tracked independently per program under a behavioral health treatment plan of care.
The combination yields roughly $263 to $318 per patient per month (G0552 contractor-priced, not included in that range). This is the program stacking ceiling. Independent time tracking is the constraint that determines whether you actually capture it.
Builder Action List: Five Product Decisions the 2026 Rule Should Trigger
Five product decisions the 2026 rule should trigger. None require new architecture from scratch; all require deliberate design choices in the data layer.
- Audit your data transmission day tracking. If the RPM platform architecture only flags whether a patient crosses the 16-day mark, the data model has a billing gap. The 2 to 15 day codes need an actual daily count with timestamps, exportable per patient per 30-day period.
- Add a 10-minute management time tier. Practices billing only under 99457 are leaving 99470 revenue on the table. The time-tracking UI should auto-suggest the correct code based on accumulated minutes, not require the clinician to know the codebook.
- Build a device clearance registry. If the platform integrates third-party devices, capture which device each patient uses and its FDA clearance status at onboarding. This is required for DMHT defensibility and good practice for RPM and RTM.
- Evaluate your audit trail for immutability. A HIPAA compliant audit log, the standard output of HIPAA compliant software development, handles data security; billing requires an append-only event ledger producing clinical evidence output a MAC auditor can verify. If billing-relevant fields are editable after the fact, the platform has an integrity problem queued up.
- Map FDA classification to DMHT eligibility before the prescribing workflow. DMHT codes require clearance for the specific condition being treated. Verify DMHT eligibility for each new indication before building the workflow that would otherwise generate non-billable documentation.
Building an RPM, RTM, or digital therapeutic platform and need to design for the 2026 codes from day one? Topflight builds billing-ready platforms with the data model, audit trail, and time tracking built in from the start.
Why Choose Topflight for Reimbursement-Enabled Digital Health Development
Topflight is among the few healthcare app developers building HIPAA-compliant digital health platforms with billing-ready architectures:
- the data transmission counting
- clinical time tracking
- interactive communication logging
- audit trail documentation that RPM, RTM, and DMHT billing requires
We track CMS rulemaking and translate it into product requirements before it becomes missed revenue or a MAC audit finding.
The RPM reimbursement 2026 changes are real, the documentation requirements are stricter, and the FDA/CMS coupling makes today’s architecture decisions tomorrow’s revenue ceiling. Talk to Topflight before you build the data model.
Frequently Asked Questions
What new CPT codes did CMS add for RPM in 2026?
CPT 99445 (RPM device supply, 2 to 15 days) and CPT 99470 (RPM treatment management, 10 to 19 minutes per calendar month). Both effective January 1, 2026.
What is CPT 99445 and when can I bill it?
CPT 99445 covers RPM device supply when a patient transmits 2 to 15 days of physiologic data within a 30-day period. Mutually exclusive with 99454.
What is CPT 99470 and how is it different from 99457?
CPT 99470 covers 10 to 19 minutes of RPM treatment management per calendar month. CPT 99457 covers 20+ minutes. They cannot be billed in the same month.
Can I bill RPM and RTM for the same patient in the same month?
No. CPT guidelines treat RPM and RTM as duplicative for the same patient in the same calendar month. Choose one based on whether monitoring is physiologic or therapeutic.
What DMHT conditions does Medicare cover in 2026?
Psychiatric disorders under 21 CFR 882.5801 and ADHD under 21 CFR 882.5803. CMS declined GI and fibromyalgia in 2026 but signaled openness for future rulemaking.